FICS Product
EDC
Electronic Data Capture that works with your workflow, not against it. AI-powered, protocol-driven, and unified with the rest of your trial.
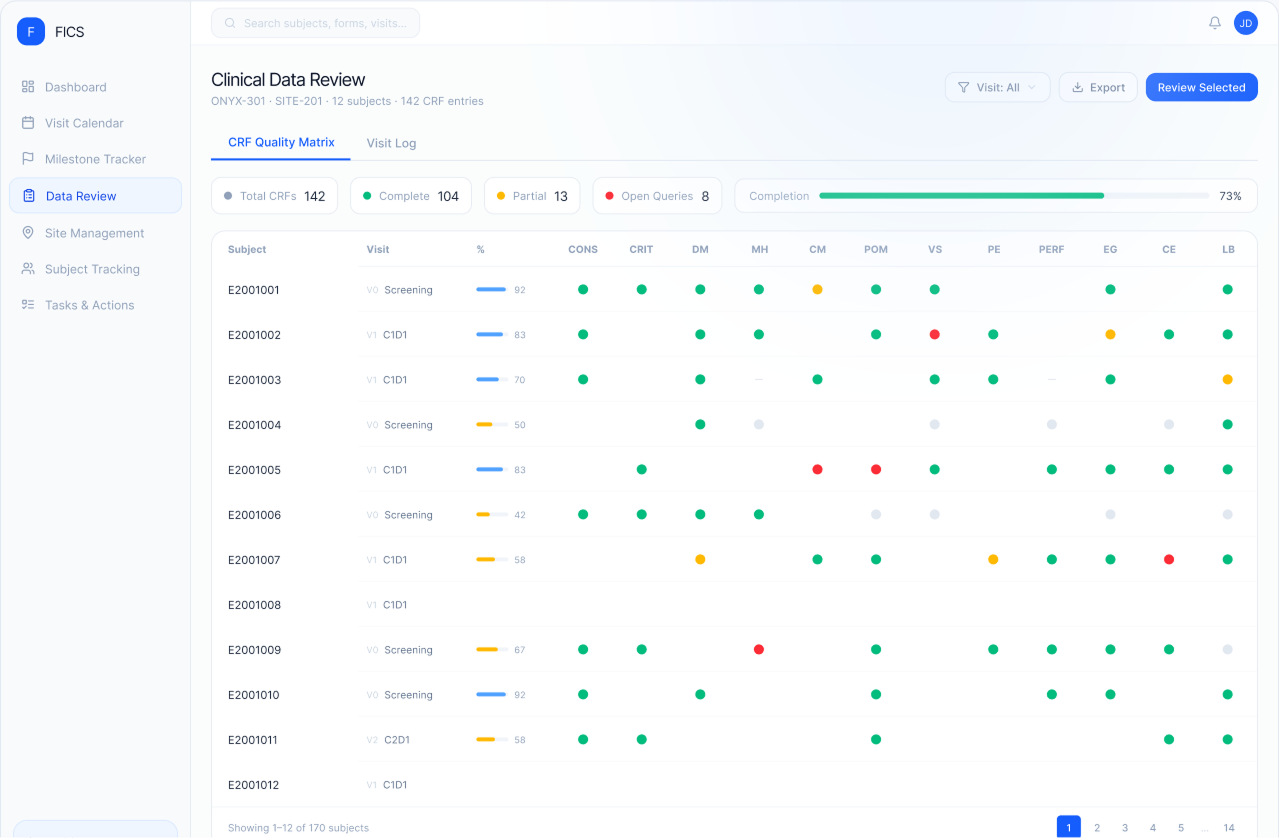
What It Is
FICS EDC is an AI-native electronic data capture system that enables protocol-driven study builds, intelligent edit checks, and real-time data validation. It captures regulatory-grade data from the point of entry and shares it instantly across the FICS platform — no exports, no reconciliation, no delays.
Who It's For
Data managers, clinical research associates, site coordinators, and biostatisticians who need clean, audit-ready data captured efficiently at the source.
Why FICS
Why It's Better with FICS
AI protocol-driven study build — dramatically accelerates form and edit check creation from protocol documents
Real-time validation at point of entry — reduces downstream queries and cleaning cycles
Natively connected to CTMS, eTMF, and safety — data entered once flows everywhere
EMR-EDC automation capable — structured and unstructured EMR data can be mapped directly into EDC forms
Built-in regulatory compliance with 21 CFR Part 11 audit trails
Faster database lock through continuous data quality monitoring
Ready for Clinical Development 2.0?
Partner with Pi Health and experience clinical trials reimagined: smarter, faster, and globally scalable.
Request a Demo